![A complex ion [Fe(H_2O)_6]^{2+} is found to be paramagnetic. Its magnetic moment is 4.89 BM. How many unpaired electrons are there? A complex ion [Fe(H_2O)_6]^{2+} is found to be paramagnetic. Its magnetic moment is 4.89 BM. How many unpaired electrons are there?](https://toppr-question-images.s3.ap-southeast-1.amazonaws.com/question_images/1560204_1727112_ans_6b23900fef074fa28cabacf524568d21.jpg)
A complex ion [Fe(H_2O)_6]^{2+} is found to be paramagnetic. Its magnetic moment is 4.89 BM. How many unpaired electrons are there?
![Explain why `[Fe(H_(2)O)_(6)]^(3+)` has magnetic moment value of 5.92 BM whereas `[Fe(CN)_(6)]^(3-)` - YouTube Explain why `[Fe(H_(2)O)_(6)]^(3+)` has magnetic moment value of 5.92 BM whereas `[Fe(CN)_(6)]^(3-)` - YouTube](https://i.ytimg.com/vi/h9yxC3M4DSM/maxresdefault.jpg)
Explain why `[Fe(H_(2)O)_(6)]^(3+)` has magnetic moment value of 5.92 BM whereas `[Fe(CN)_(6)]^(3-)` - YouTube
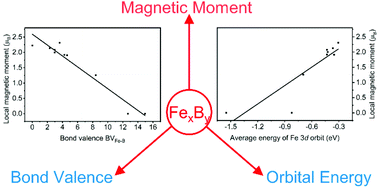
Insight into the magnetic moment of iron borides: theoretical consideration from the local coordinative and electronic environment - Dalton Transactions (RSC Publishing)
What will be the theoretical value of spin only magnetic field when Fe(SCN)3 reacts with the solution containing F ions to yield a complex
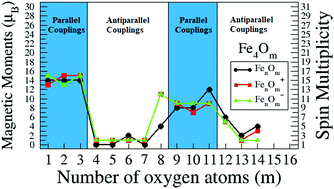
Relation between structural patterns and magnetism in small iron oxide clusters: reentrance of the magnetic moment at high oxidation ratios - Physical Chemistry Chemical Physics (RSC Publishing)

a) Magnetic moments of Fe atoms calculated from first-principles at... | Download Scientific Diagram
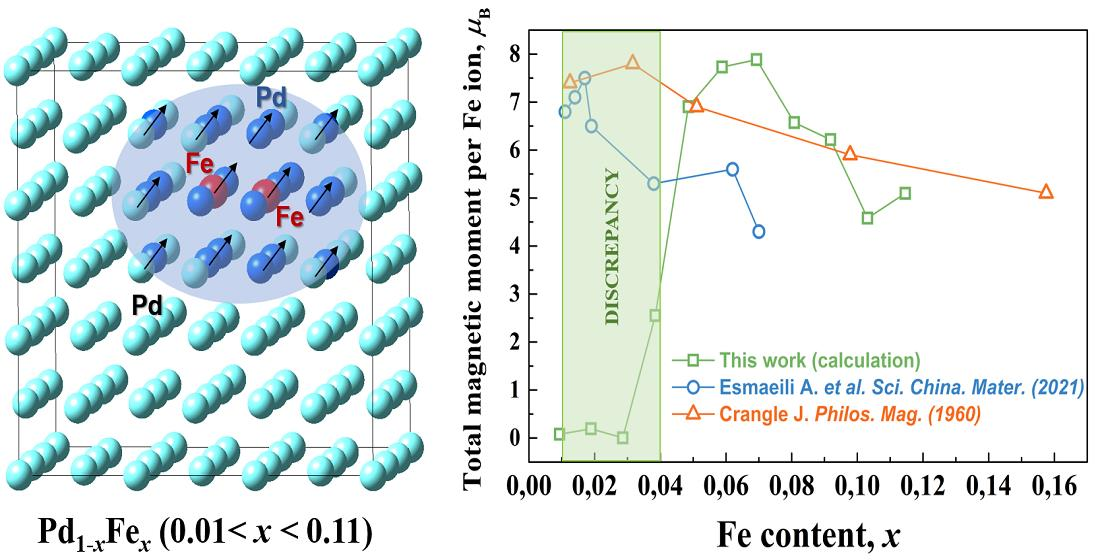
Crystals | Free Full-Text | Ab initio Investigation of Impurity Ferromagnetism in the Pd1−xFex Alloys: Concentration and Position Dependences
20.Among venedium, chromium, mengenize,and Fe which will have the highest magnetic moment ? A) v B) cr C) Mn D) Fe

![What is the magnetic moment Fe^{3+} ion in [Fe(CN)_6]^{3-}? What is the magnetic moment Fe^{3+} ion in [Fe(CN)_6]^{3-}?](https://haygot.s3.amazonaws.com/questions/1840618_1731530_ans_30dc94e7428a4b59a655c78fb7cf88bc.jpg)



![The spin magnetic moment of iron in `K_(3)[Fe(CN)_(6)]` - YouTube The spin magnetic moment of iron in `K_(3)[Fe(CN)_(6)]` - YouTube](https://i.ytimg.com/vi/m2ePFFONgdA/mqdefault.jpg)